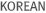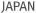Consulting
HOME > Consulting > Global Regulatory Affairs > GHS & (M)SDS Authoring
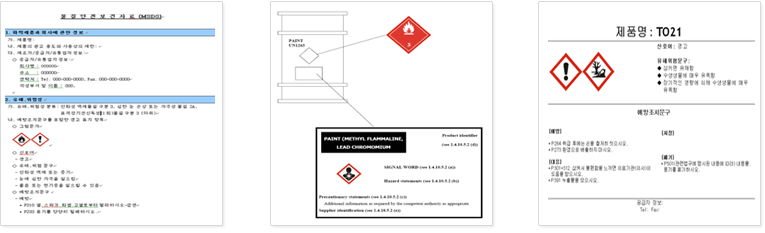
GHS & (M)SDS Authoringt
We provide overall chemical management services such as Regulatory Affairs,
Risk Assessment, Emission Management, etc.
Risk Assessment, Emission Management, etc.
- GHS & (M)SDS Authoring
Overview
We author and handle (M)SDS under GHS and various international regulations and consult on chemical classification and labeling from the beginning of manufacture.
Our core services include the following
- GHS Labeling and (M)SDS authoring/verification under Chemical Substance Control Act, Occupational Health and Safety Act, Control of Dangerous Substances Act
- GHS & (M)SDS Authoring under international regulation (EU CLP, China Chemical Inspection and Regulation Service(CIRS), Japan Industrial Safety and Health Law (ISHL), etc)
- Multilingual Services (English, Japanese, Chinese)
- SMART MSDS™ solution provisioning (TO21’s self-developed)